How to Use Colorimeter?
Colors are an integral part of our daily experiences, influencing our emotions, actions, and beliefs, whether we're aware of it or not. They have the power to evoke feelings, convey warnings, provide essential information, and bring about joy. Despite their omnipresence, describing colors proves challenging. This difficulty arises from the fact that individuals perceive colors differently, and the sheer multitude of colors that the human eye can detect lacks a comprehensive set of descriptions.
A colorimeter is a device used to measure the absorbance or transmittance of light by a solution or sample, typically in chemistry and biology applications. It is commonly used to determine the concentration of a substance in a solution based on its color intensity. ATO online shop will go into detail about the general guide on how to use a colorimeter.
 Working Principle
Working Principle
At its most basic, a colorimeter works by passing light of a particular wavelength through a solution and then measuring the light on the other side. In most cases, the thicker the solution, the more light is absorbed, as can be seen in the difference between the light from the source and the light passing through the solution. To find the concentration of the unknown sample, several solution samples of known concentrations are first prepared and tested. They are then plotted on a chart to produce a calibration curve. When testing an unknown sample, the results are compared with the known sample on the curve to determine the concentration.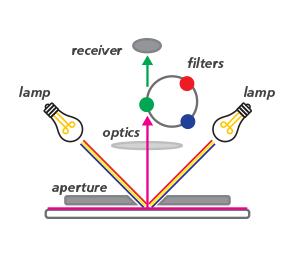
Prepare the Sample
Ensure that your sample is properly prepared according to the specific requirements of your experiment or analysis. Before using the colorimeter, it's essential to calibrate it with a blank or reference solution. This establishes a baseline for the measurements. Insert the blank solution into the colorimeter and set it to zero absorbance or 100% transmittance. Choose the appropriate wavelength of light for your analysis, as different substances absorb light at different wavelengths. Adjust the colorimeter to the desired wavelength using the wavelength selection knob or digital display. Place your sample into the colorimeter, ensuring homogeneity and eliminating air bubbles that could affect the readings.
Measure Absorbance or Transmittance
To measure absorbance or transmittance, close the lid of the colorimeter to prevent external light interference. Record the absorbance or transmittance value displayed on the colorimeter, as it is proportional to the concentration of the substance in the sample.
Data Analysis and Clean
For data analysis, use the recorded data to calculate the concentration of the substance. This often involves comparing absorbance or transmittance values to a standard curve or known calibration curve. After each measurement, thoroughly clean the cuvette or sample holder to prevent contamination between samples. Follow the manufacturer's instructions for proper maintenance and cleaning of the colorimeter. Turn off the colorimeter and disconnect it from the power source when finished with measurements.

Interpret Results
Analyze and interpret your results based on the objectives of your experiment. Compare your measurements with known standards or control samples to validate your data.
The basic principle behind colorimetry is that the color of a substance is related to its chemical composition and concentration. By measuring the absorbance or transmittance of light at specific wavelengths, scientists and researchers can obtain quantitative data about the concentration of a particular substance in a sample. Colorimeters find applications in various industries, including environmental monitoring, food and beverage analysis, clinical chemistry, and water quality testing. They are valuable tools for researchers and analysts who need to perform rapid and accurate color-based analyses in their work.

